Enhanced Games Bounces Big from Extreme Low Like NIA Predicted
On May 14th, NIA sent out an alert discussing Apeiron Investment Group's latest venture, the…
NIA’s QIMC Gains 8.62%, Eric Sprott’s MAXX Declines 1.29%
Over the weekend, NIA highlighted the strength of our stock suggestions discussed in our March…
Billionaires Investing Big into Scottish Football
Why Celtic plc Could Be the Next Major Takeover Target in Scottish Football Back in…
How Is Eric Sprott’s HYMC Doing?
On March 19th, NIA sent out an alert entitled, 'Eric Sprott Was Buying HYMC at…
Celtic Hire Martin O’Neill as Permanent Manager
In recent weeks, NIA has been saying that Celtic plc (LSE: CCP), or CLTFF on…
QIMC No Longer High Risk Like Before?
We already knew last fall that Eric Sprott was backing Max Power Mining (CSE: MAXX)…
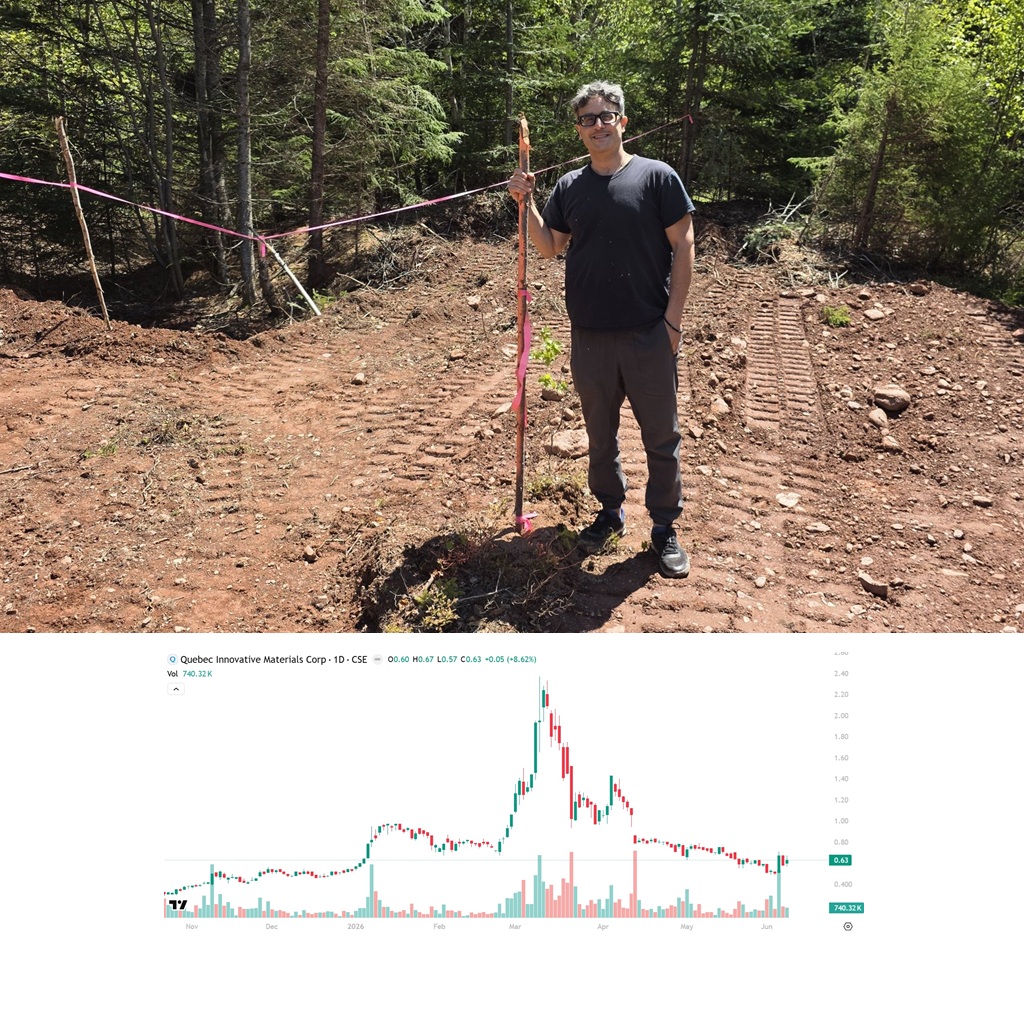
QIMC Jumps 32.67% Following Presentation to Quebec National Assembly
QI Materials (CSE: QIMC) gained by 32.67% today to $0.67 per share after delivering a…
QIMC Recognized by Quebec Lawmakers as Natural Hydrogen Industry Leader
QIMC Appears Before Quebec National Assembly on Bill 17, Highlighting Quebec's Natural Hydrogen Potential Natural…
MANU Up 11.84% to $23.61 and World Cup Begins in One Week
On May 18th, NIA predicted with Manchester United (MANU) at $19.81 per share that it…
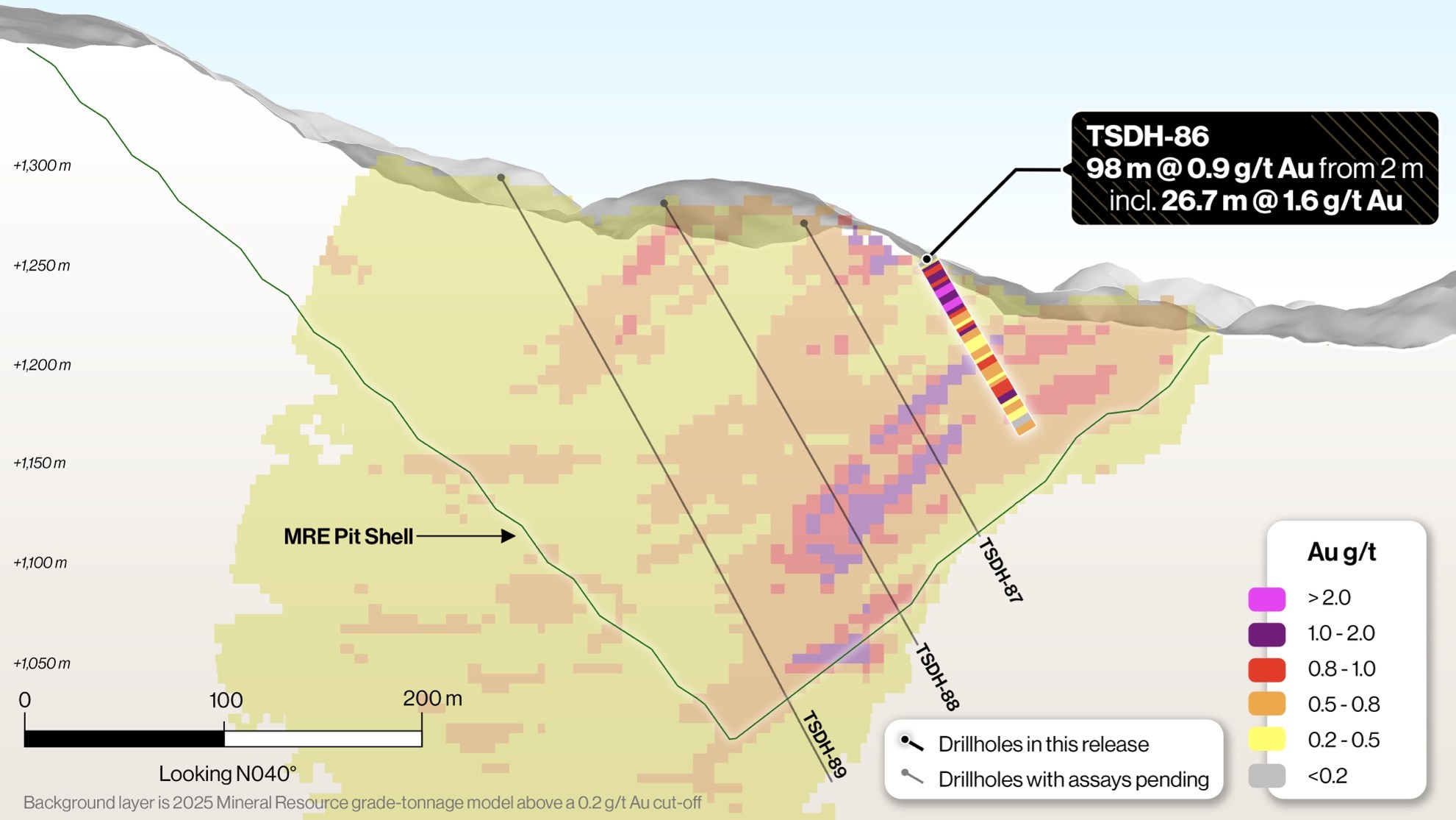
Tiger Gold Hits 98m of Gold Near Surface and Ends Hole in Mineralization
Tiger Gold Intersects 98 m @ 0.9 g/t Au including 26.7 m @ 1.6 g/t…